Editor’s Note: This story contains graphic images.
CNN —
A rare strain of bacteria found in recalled eye drops has been linked to dozens of infections, as well as cases of vision loss and surgical removal of eyes. This week, the US Centers for Disease Control and Prevention reported two additional deaths – a total of three – have been linked to the bacterial infection.
Global Pharma Healthcare recalled its Artificial Tears Lubricant Eye Drops that were distributed by EzriCare and Delsam Pharma last month, and the agency has been warning people not to use the drops.
As of March 21, the CDC has identified 68 people in 16 states with infections of a rare strain of drug-resistant Pseudomonas aeruginosa never before reported in the United States. Most of the patients reported using artificial tears, the CDC said, and EzriCare Artificial Tears was the brand most commonly reported.
Reported adverse events include infections of the cornea, bloodstream, respiratory tract and urinary tract. There are eight reports of lost vision and four reports of surgically removed eyeballs.
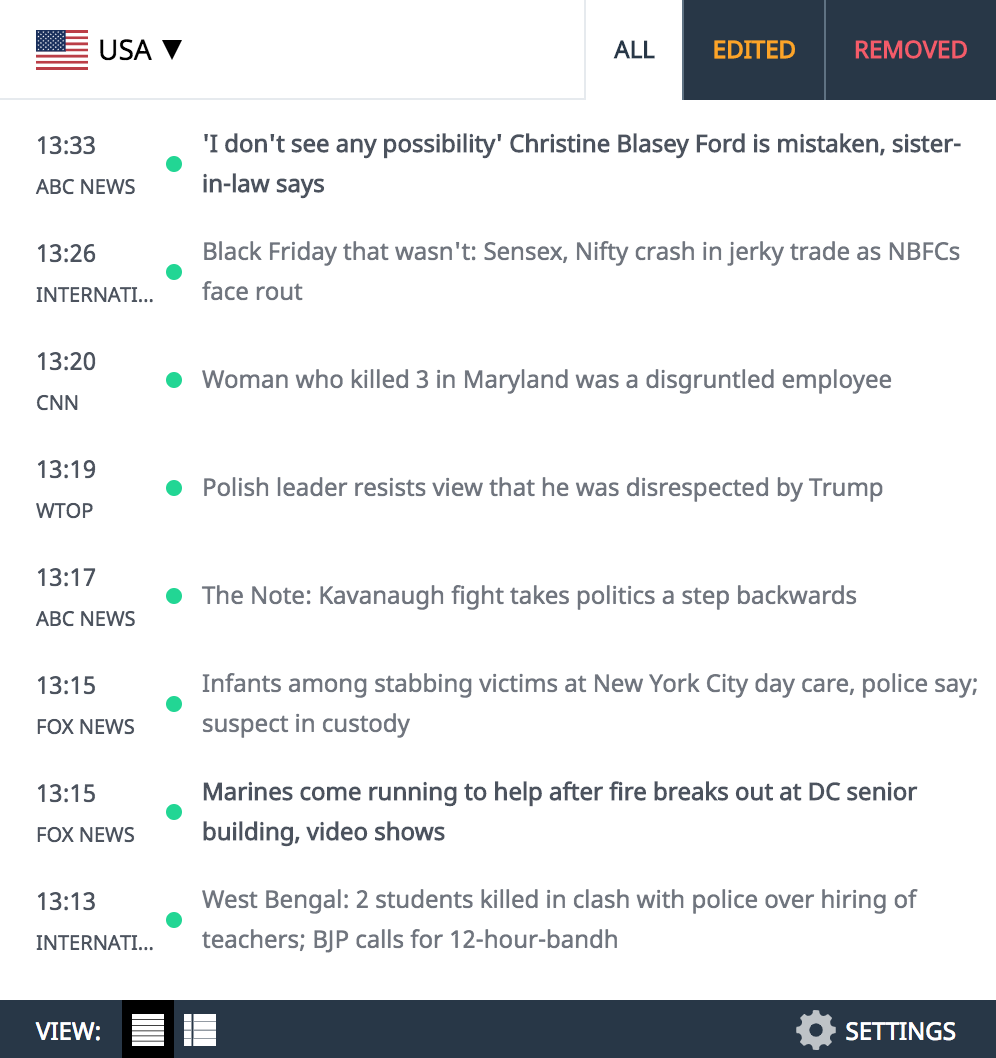
One of the new case reports described a 72-year-old woman who lost vision in her left eye after using EzriCare artificial tears for dry eyes for about a week.
“She started noticing some blurry vision in her left eye for a few days,” said Dr. Ahmed Omar, an ophthalmologist at University Hospitals Cleveland Medical Center, who treated the woman. “It was initially painless, but according to the patient and her husband, one morning she woke up and she had a yellow discharge on her pillow. And that’s when she started noticing that the appearance of her eye had changed.”
The woman went to the emergency room, where doctors discovered a large ulcer on her left cornea, nearly involving the entire eye. She was admitted to the hospital for three weeks, requiring IV antibiotics, antibiotic eye drops and multiple surgical interventions.
The woman’s case was complicated by a serous choroidal detachment, an abnormal accumulation of fluid, which ultimately led to vision loss in the left eye.
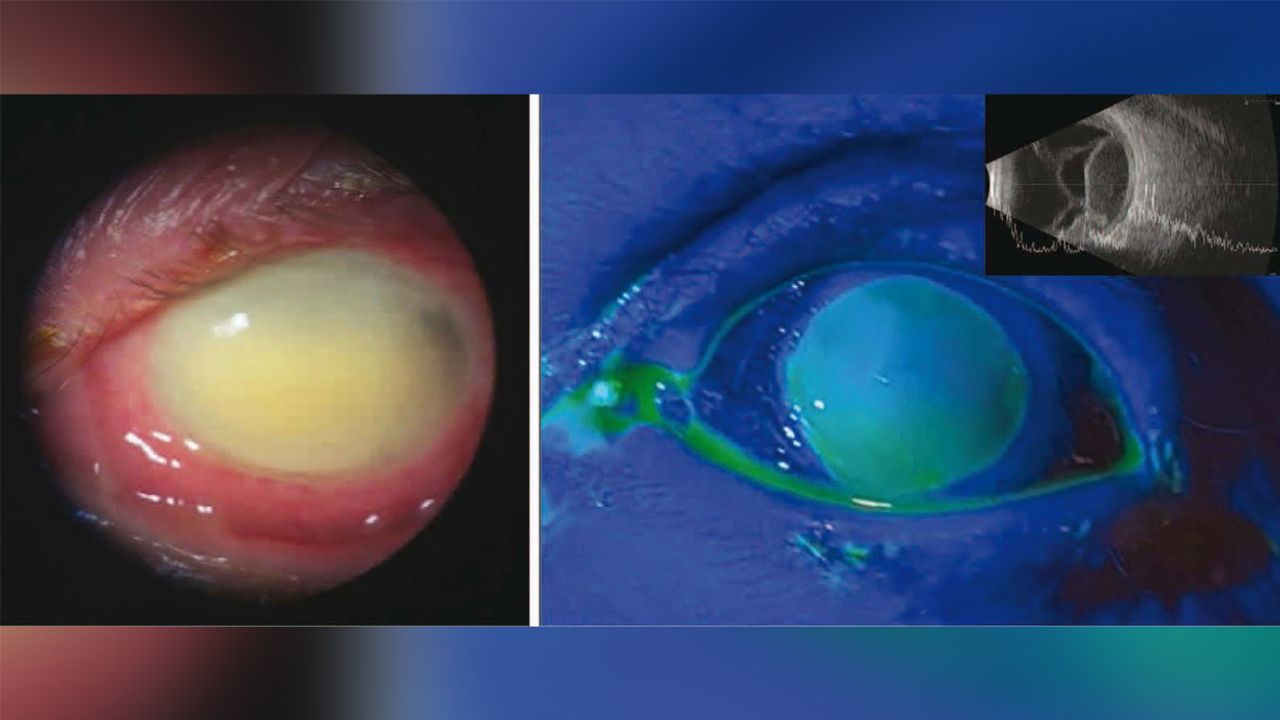
Another case involved a 72-year-old man who developed multidrug-resistant Pseudomonas aeruginosa keratitis, an infection of the cornea.
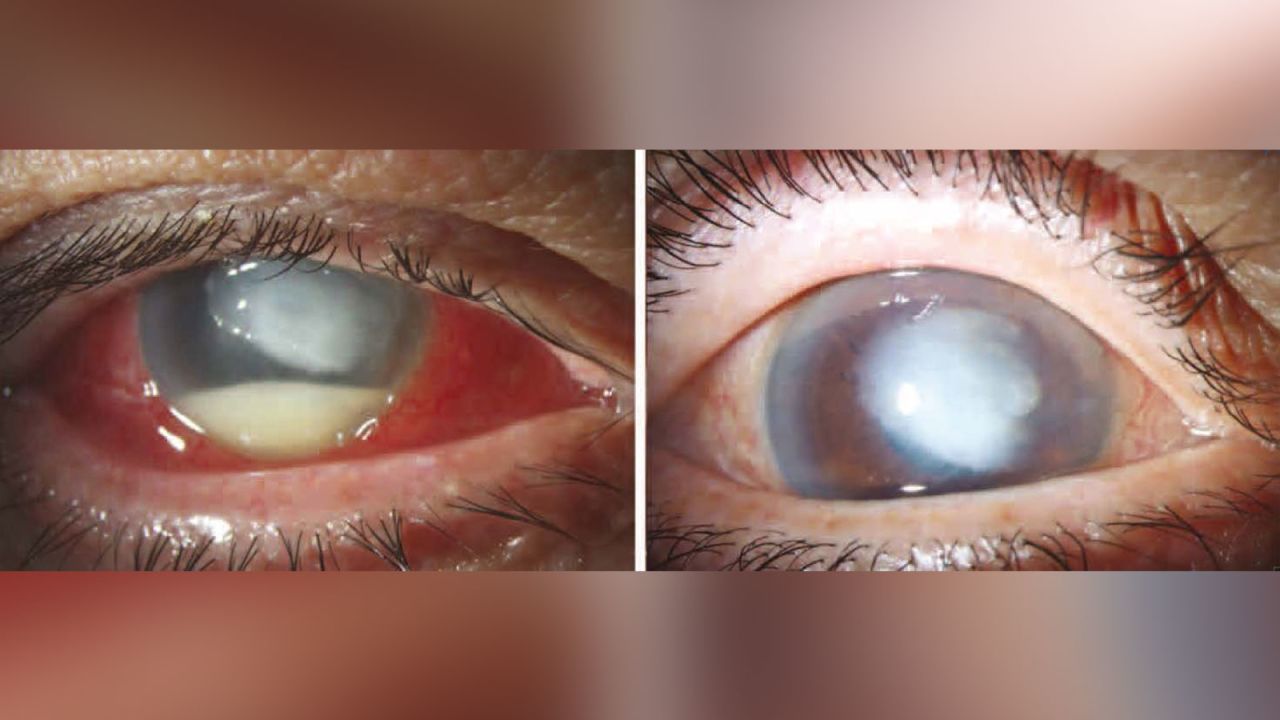
The man went to the Bascom Palmer Eye Institute in Miami after a day of severe pain and decreased vision in his right eye. He had no previous eye problems but said he had used EzriCare artificial tears for eye dryness.
“When we examined his right eye, there was a severe corneal infection,” said Dr. Marissa Shoji, a resident physician at the institute, who treated the patient. “He could only see shadows and was not able to see letters due to the extent of the ulcer.”
The man was started on strong antibiotics, said Dr. Naomi Gutkind, a resident physician who also treated him.
“We typically expect some degree of improvement these medications, but when we saw him two day later, he was getting far worse,” she said. “So that’s when we inquired about specifically the EzriCare tears, because we knew they were associated with resistant infection that may not respond to those really strong antibiotics.”
Cultures from the man’s cornea and EzriCare bottle grew the same strain of multidrug-resistant Pseudomonas.
The man’s vision at his two months follow-up appointment was 20/400, meaning he can see at 20 feet what healthy people can see at 400 feet.
“At some point, he was in danger of having permanent vision loss,” said Dr. Guillermo Amescua, an ophthalmologist at Bascom Palmer Eye Institute. “He now has what is called corneal blindness because he’s 20/400 and has a corneal scar, but with corneal transplantation, he might have a better prognosis.”
Shoji says this case highlights the short-term and long-term effects of Pseudomonas keratitis. In the short term, people may have terrible pain, decreased vision and a risk of a hole in the eye that could spread infection.
“In the long term, even after the infection is cleared, there’s a risk of needing a surgery such as a corneal transplant or other different types of surgeries to address scarring that may also significantly limit the vision,” she said.
EzriCare said in a statement that after learning about the CDC’s investigation of Pseudomonas infections on January 20, it “immediately took action to stop any further distribution or sale of EzriCare Artificial Tears. To the greatest extent possible, we have been contacting customers to advise them against continued use of the product.”
The physicians involved in the new studies hope that by highlighting these cases, they can spare others from similar problems.
Shoji advises against using EzriCare or Delsam eye drops while the investigation is ongoing.
“If you have any eye symptoms, things like eye pain, eye redness, decreased vision, please see an ophthalmologist for evaluation,” she said. “And when you come, please bring anything that could potentially be causing this infection, especially eye drops, contact lenses or contact lens cases.”
The CDC says symptoms can include yellow, green or clear discharge from the eye; eye pain or discomfort; redness of the eye or eyelid; feeling like something is in the eye; increased light sensitivity; and blurry vision.
Doctors urge everyone to use caution with eye drops.
“I think a lot of times, people don’t think of eye drops or even things like contact lenses as medical devices. I think they think of them more as kind of just an over-the-counter product like a skin lotion or something like that,” said Dr. Christina Prescott, a NYU ophthalmologist who wrote a commentary published alongiside the case reports on the potential rare dangers of presumably benign artificial tears.
Prescott says people should keep eye drops sterile. That means avoiding touching the tip of the bottle with their hands, skin or eye; not sharing the bottle with someone else; and not using expired products.
People should also use caution with preservative-free eye drops because contamination can lead to serious infection.
“Anything you’re putting in your eye, if it’s contaminated, is going to have a high risk of infection just because your eyes are so vulnerable,” she said.